|
While the electrons that are present in the surrounding orbits are very light. The mass of protons and neutrons is the same and it is 1.6749 × 10 -27 kg. Protons and neutrons are the heavy particles in an atom. :max_bytes(150000):strip_icc()/PeriodicTableCharge-BW-56a12db13df78cf772682c34.png)
The mass of an atom completely depends on the number of protons and number of neutrons present in the nucleus of an atom. Due to this, the total mass of hydrogen atom is the least of all known elements. Hydrogen is the lightest gas because it consists of only one proton and one electron. Electronic configuration of first 30 elements of Periodic table. Detailed Periodic table with electron configuration (Image).Ģ). Well, in the modern printed periodic tables, you can see the Hydrogen element being placed in the group 1 along with the alkali metals.Īpart from having similar properties as that of group 1 and group 17 elements, hydrogen element is placed in 1st group because it has similar outermost electron configuration as that of alkali metals (i.e ns 1).ġ). So where should hydrogen be placed? In group 1 or group 17? So in short, I simply want to say that properties of hydrogen are similar to group 1 elements as well as group 17 elements. Similarly hydrogen also exists as a diatomic molecule (i.e H 2). Hydrogen also forms oxide (i.e H 2O or dihydrogen oxide.)Īlkali metals form sulphides (like Na 2S, K 2S, etc) and hydrogen also forms sulphide (i.e H 2S).Īlkaline metals form halides (like NaCl, KCl, etc) and hydrogen also forms halide (i.e HCl).Īpart from showing similar characteristics like that of group 1 metals, hydrogen also shows similar characteristics as that of halogens (group 17 elements).įor example halogens have very high ionization enthalpy, and hydrogen also have very high ionization enthalpy.Īlso, the halogens exist as diatomic molecules (like F 2, Cl 2, etc). The same chemical property of oxide formation is found in Hydrogen also. HEX (Hexagonal) Melting point 13.99 K or -259.16 ☌ or -434.49 ☏ Boiling point 20.271 K or -252.87 ☌ or -423.18 ☏ Density 0.0898 g/L Main isotope 1H, 2H Who discovered Hydrogen and when?įor example, group 1 elements form oxides like lithium oxide, sodium oxide, potassium oxide, etc. 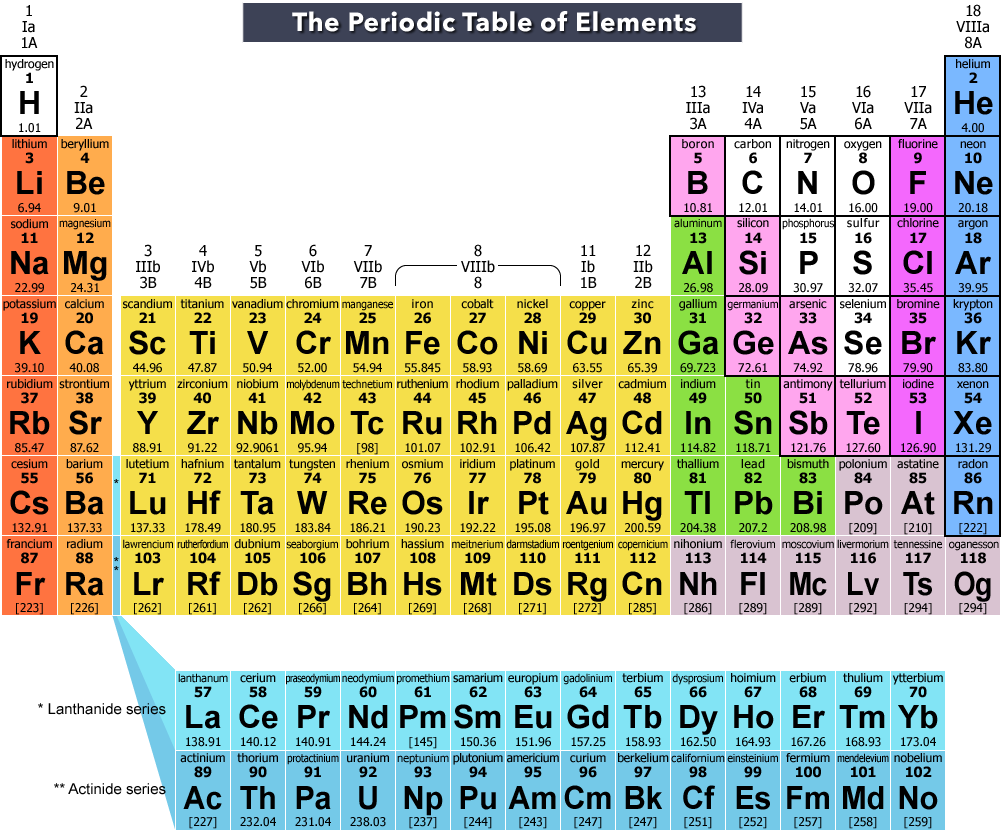
Protons 1 Neutrons 0 Electrons 1 Symbol H Atomic massġ Electronic configuration 1s 1 Atomic radiusġ20 picometers (van der Waals radius) Valence electronsġ 1st Ionization energy 13.598 eV Electronegativity Group: 1, Period: 1, Block: s Category Other Nonmetals Atomic number or Hydrogen Element (H) Information Appearance Colorless gas State (at STP) Gas Position in Periodic table So if you want to know anything about hydrogen element, then this guide is for you. In fact, the table mentioned below is the perfect information box (Which gives you every single detail about the Hydrogen element in Periodic table.) 
This is a SUPER easy guide on hydrogen element.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
